NMR spectra and relaxation times T1 and T2 for 31P in membranes of Rhodobacter sphaeroides were investigated at different relative humidity levels. The results are compared to the hydration curves, fatty acid composition and the structure-dynamic and functional characteristics of the membranes of photosynthetic bacteria Rb. sphaeroides, Rhodospirillum rubrum and Ectothiorhodospira shaposhnikovii. The differences in the state of lipid phase of these membranes are revealed under low humidity, and this is conducive to variability of their structural dynamic and functional characteristics during the hydration process. Based on the results obtained and the data on model systems, four stages of hydration process are distinguished with different effects on the structure and dynamics of membrane components. These stages are: hydration of a portion of polar groups, involvement of water molecules in the hydrogen bonds within macromolecules and the lipid phase, hydration of all polar groups with the appearance of water with high dielectric constant thus making possible the lateral diffusion within the membrane and realization, through water participation, of conditions within organelles and cells required for the process regulation at these levels. The mechanism of water action on various membrane components and their dynamics at each stage are discussed, as well as the effect of different types of motion on the efficiency and regulation of electron transport in the photosynthetic chain of the membranes studied.
(Received 10 September, 1996)
Investigation of the hydration effects on photosynthetic membranes (chromatophores) in our earlier studies by 1H, 13C and 31P NMR spectroscopy revealed a crucial role of intramembrane motion in achieving high efficiency of processes involved in the electron transport chain [1-4]. These results also allowed differentiation of three definite motion types with clearly distinct frequencies associated with different structures and responsible for such processes in various chain sites [3, 4]. At the same time, the possibility of direct comparison of functional parameters with the data on structure and dynamics of such membranes appears to be interesting for a wider range of problems.
The independence of measured parameters of the diffusional restrictions in the reaction with substrate makes it possible to derive direct data on the role of inner motion in biopolymers and membranes in providing the high efficiency of biological reactions. Furthermore, the occurrence of stages in the electron transfer chain, which are largely different by their time characteristics, allows comparison of their times with those of structural rearrangements within the volumes increasing in accordance with the slowing-down of the rates of electron transfer stages. The character of occurring changes should reflect the status of membranes, organelles or the entire cell, and this may form the basis for the regulation ensuring both the most efficient use of electron flux in the photosynthesis chain and the prevention of structural damage under the conditions of excess electron flux not involved in subsequent biochemical reactions. The medium restructuring per se may in turn be a factor reflecting the sensitivity of biological systems to weak volume-distributed exposures.
Finally, photosynthetic membranes are convenient models for studying general mechanisms of water interaction with real multicomponent biological membranes. Basic studies of the hydration effect on the membrane structure and dynamics were carried out on purely lipid model systems or those including only individual components of different nature. This is far from reflecting the behaviour of real membranes in which the number of different lipids is as high as a few hundreds or even thousands [5], let alone other components. In this respect, highly interesting seems to be the variability of photosynthetic membrane composition and structure in various species of purple bacteria allowing apparently both the differentiation of specific systems responsible for functional or structural changes and elucidation of their molecular mechanisms. Certain possibilities are opened by the use of 13C and 31P NMR spectroscopy allowing separate investigation of structural-dynamic characteristics of the protein component as well as the polar and nonpolar moieties of the lipid component of membranes in the course of their hydration in the solid phase [3, 4].
Our previous studies of photosynthetic membranes of such purple bacteria as Rhodospirillum rubrum, Ectothiorhodospira shaposhnikovii and Rhodobacter sphaeroides revealed noticeable differences in the water action on their structural-dynamic and functional characteristics [1-4]. This, however, raises a number of questions, specifically on the relationship between the dynamics and the efficiency of electron transfer in the system of quinone acceptors in membranes of Rb. sphaeroides and E. shaposhnikovii (due to its maintenance at a high level in dried preparations) [2]. A possible explanation is associated with differing structural states of specific membranes, as indicated by comparison of relaxation times T1 and T2 of 31P nuclei. In the R. rubrum membranes characterized by the predominance of phosphatidylethanolamine (PE) in their lipid phase [6], measurements of T1 [3,4] revealed a paradoxical dependence - the first humidification stages led to the strengthening but not weakening of bonds in the polar region of membranes. This is hardly consistent with the presence in dried membranes of a hexagonal phase for the lipids whose polar groups are in direct contact. However, the use of standard technique for evaluating the phase state of lipids - from the curve shape for 31P nuclei [7, 8] - failed to provide any unequivocal answer due to the multicomponent character of this system. On the other hand, the data on T1 from 31P nuclei in R. rubrum membranes differ noticeably from those for Rb. sphaeroides membranes [4] obtained at three humidity levels. They also differ from the results obtained on model systems indicating that PE tends to form a hexagonal phase, though different from that of PE isolated from different objects [7, 8].
The above considerations formed the basis for more thorough comparative studies of the structural-functional parameters of membranes of purple bacteria Rb. sphaeroides and R. rubrum at different humidity levels by 31P NMR spectroscopy and for their comparison with other structural and functional parameters. Combined with the earlier data, including those for E. shaposhnikovii membranes [2], this approach allows a deeper insight into the effect of water on the structure, dynamics and different functional characteristics of photosynthetic membranes and their components.
EXPERIMENTAL
Non-sulfur purple bacteria (Rhodobacter sphaeroides and
Rhodospirillum rubrum) and sulfur bacteria (Ectothiorhodospira
shaposhnikovii) were cultured for 3-5 days under anaerobic
conditions in a luminostat at 30°C. Cells of R. rubrum
and Rb. sphaeroides were grown on Ormerod's medium, cells
of E. shaposhnikovii were cultured on Larsen's medium [9].
Photosynthetic membranes (chromatophores) were isolated from bacterial
cells using a standard technique [10]. The cells separated from
culture medium were suspended in 0.05 M Tris-HCI buffer, pH 6.8-7.0
(pH 8.0 in the case of E. shaposhnikovii), containing 0.05
M sucrose (0.25 M sucrose in the case of E shaposhnikovii)
and 0.005 M MgSO4 Cells were destroyed by sonication
(20-22 kHz, 0.5 A) for 3-5 min. After removal of intact cells
and large particles by centrifugation the chromatophore fraction
from the separated supernatant was sedimented at 100,000-150,000
g for 1-2 h at 0-4°C.

Figure 1. Membrane hydration isotherms for different photosynthetic bacteria, g H2O per g dry sample mass. a, Rhodospirillum rubrum (1) Rhodobacter sphaeroides (2); b, Ectothiophodospira shaposhnikovii with adsorption of H2O (3) and D2O (4). Weight values for D2O are multiplayed by 0.9, a proportionality coefficient for molecular masses of H2O and D2O .
Figure 2. NMR spectra for 31P
nuclei in membranes of Rhodobacter sphaeroides (a) and
Rhodospirillum rubrum (b) at different values of relative
humidity P/P0 .
Chromatophores intended for NMR measurements were subjected to liophilic drying under a reduced pressure of 10-3 mm Hg with subsequent moistening in a desiccator over saturated saline solutions to a definite degree of their hydration [11]. The electron-transport activity of R. rubrum and Rb. sphaeroides chromatophores as a function of their hydration extent was investigated by the method of differential spectroscopy on film preparations obtained by drying on glass supports either the initial suspension of chromatophores or a moistened liophilized powder used in NMR studies. Films of E. shaposhnikovii chromatophores which preserve the cytochrome activity were prepared as in [12]. The extent of sample hydration was determined by the gravimetric method and also by direct titration with Fischer's reagent using the electrometric technique of equivalency point recording [13].
Lipids were extracted from photosynthetic membranes on cold successively with isopropanol, its mixture with chloroform (1:1) and twice with a mixture of chloroform and methanol [14]. The extraction completeness was estimated from the level of fatty acids in the sediment by the method of inner standard after the fourth extraction [15]. The combined extracts were evaporated in a rotary drier at 40°C. Methyl esters of fatty acids (MEFA) were obtained by alkaline hydrolysis of lipids with NaOH and methylation of fatty acids in a mixture of methanol with etherized boron trifluoride [16]. MEFA were separated in a Chrom-52 chromatograph (Laboratorni Pristroje, Czech Republic) on a column (2.5 x 3 mm) packed with 3% 1,4-butanediol succinate on Super-lnerton (0.160-0.200 mm) (Lachema, Czech Republic). The column was maintained at 195°C; the flame-ionizing detector and evaporator, at 210°C. MEFA were identified using standards of saturated and unsaturated acids (Sigma, USA) and known values of their relative retained volumes [17, 18]. The peak areas were measured with a Cl-100 electron integrator (Laboratorni Pristroje).
The NMR spectra of 31P nuclei and their relaxation times T1 and T2 were recorded on a CXP-300 Bruker spectrometer at 121 MHz. The spin-lattice relaxation time T1 was measured using a "saturation-recovery" sequence [19] of a series of 90° pulses with interpulse interval t = 0.01, 0.02, 0.04, 0.08, 0.15, 0.30, 1.20, 2.40, 4.50, 8.00, 15.00, 25.00, 40.00 s. The complex curves of longitudinal magnetization recovery for 31P nuclei were approximated as the sum of two exponents ("fast" and "slow") with concomitant determination of T1 and their relative shares using the EXPO program. Similar procedure was performed to estimate the amplitude values of curves and the integral areas under the curves using not less than 200 accumulations. The spin-spin relaxation time T2 (more strictly, T2* < T2 due to additional widening of resonance associated with anisotropy of chemical shifts for 31P nuclei in these samples) was measured directly from the resonance half-width and used solely for relative estimates. In this case, each curve was the mean of not less than 1000 measurements.
RESULTS AND DISCUSSION
Comparison of bacterial membrane hydration curves for R. rubrum, Rb. sphaeroides and E. shaposhnikovii revealed substantial differences (Fig. 1). Most specific are the hydration curves of R. rubrum membranes exhibiting distinctly a strong increment of water sorption in the interval P/Po=0.3-0.5, whereas the further humidity increase up to P/Po=0.80 is not virtually conducive to any noticeable rise in the membrane hydration extent. This effect was found to be more dramatic in the protein-pigment complexes of reaction centres (RC) isolated from such membranes using lauryidimethylamine oxide (LDAO) [1], a detergent capable of forming (like PE) hydrogen bonds between its molecules.
The membrane hydration curves of Rb. sphaeroides are smoother that those of R. rubrum which are also characterized by a definite sorption water increment in this interval. At the same time, the bacterial membranes of E. shaposhnikovii, containing the peripheral protein cytochrome c (immediate electron donor for photoactive bacteriochlorophyll), differ from two other bacterial membranes by the shift of water absorption increment region toward somewhat higher P/Po values; at high humidity a further more significant increase in the water adsorption is registered. At low and intermediate humidity levels, these membranes also reveal a noticeable difference in the H2O and D2O adsorption (regarding the number of sorbed molecules) which eventually disappears with further humidity growth [2] (Fig. 1b).
In addition to generally noticeable differences between the membrane hydration curves of R. rubrum and E. shaposhnikovii, their membranes display, along with a considerable increment of sorbed water, another common phenomenon consisting in a substantial increase in the mobility of molecules and their specific groups, besides water, and recorded from changes in T1 and T2 values for protons [1, 2], and also T1 for 13C nuclei in the case of . rubrum [3, 4]. Comparison of T2values for protons of non-water molecules suggests that at low humidity the membranes of E. shaposhnikovii contain molecules and molecular groups with a mobility one order of magnitude higher than that in the R. rubrum membranes [1, 2]. Such mobile groups are primarily the tails of lipid molecules.
The differences observed in the mobility of lipid tails cannot
be associated with the fatty acid composition of membranes as
indicated by the results of its determination in these species
of photosynthetic bacteria and in the Chromatium minutissimum
(Table 1). The proportion of long-chain molecules seems to be
even higher in Rb. sphaeroides than in R.. rubrum
as suggested by the literature data [20]. Slight differences in
the summary ratio of saturated and unsaturated lipids in these
membranes are likewise inadequate for explaining the differences
in mobility, the more so that in the E. shaposhnikovii
membranes with maximal levels of saturated lipids the mobility
of tail lipids at low humidity remains relatively high.
Additional information about changes occurring in membranes in the course of hydration is provided by the evaluation of the phase state of such membranes. To this end, as noted above, the form of the resonance curve for the phosphorus nuclei is used [7, 8]. As one can see in Fig. 2, in contrast to the earlier curves for the R. rubrum membranes shown for comparison, at low P/Po values of 0.12 and 0.29 the shape of resonance curves for 31P in the membranes of Rb. sphaeroides has a clear-cut left-side asymmetry that may be related to the occurrence of hexagonal phase in lipids [7, 8]. This situation, when the polar groups of lipids are in direct contact, requires the existence of relatively rigid intermolecular bonds and low mobility of polar lipid groups. The latter was characterized using the spin-lattice relaxation time T1 for the phosphorus nuclei. According to the NMR theory [21], the increasing mobility in the range of low frequency motion (below the resonance frequency), e.g. due to a weakening rigidity of intermolecular bonds in the course of membrane hydration, must induce transition from high T1 values characteristic of low-mobile structure to the smaller ones. Such a pattern is in fact observed for the membranes of Rb. sphaeroides bacteria (Table 2). In this case, like in R. rubrum, with a certain approximation two groups of phosphorus nuclei may be distinguished which appear to belong to different sites with noticeably different mobilities within the limits of polar region of membranes. In this situation, the decrease of T1 values with the growing humidity is associated primarily with the slow component of longitudinal magnetization recovery for the nuclei of phosphorus atoms which belong to the more rigidly bound lipid molecules and account for their major mass. The characteristics of a more mobile and quantitatively smaller portion of phospholipid molecules display relatively small changes at the two humidity levels indicated above (Table 2).
At the next hydration stage (in the range of P/Po= 0.29-0.43),
where noticeable changes in the structural-dynamic characteristics
of bacterial membranes were recorded in R. rubrum and E.
shaposhnikovii, the resonance asymmetry for the phosphorus
nuclei in the Rb. sphaeroides membranes becomes less pronounced
that is in general consistent with the suggestion ol the
disappearance of at least a portion of hexagonal phase within
the membrane. According to the measurements of T1 for
the phosphorus nuclei (Table 2), the structural rearrangement
is accompanied by the further mobility increase in the polar region
of lipid molecules with the invariability of the ratio between
the components corresponding to the shares of more and less mobile
molecules. Finally, a further increase of relative humidity P/Po
to 0.76 and above induces a noticeable resonance narrowing with
the disappearance of its asymmetry (Fig. 2). The share of more
mobile molecules increases dramatically in the polar membrane
region, and there appears a narrow band characterized by a 10
times higher value of T2 = 200 s (Table 2) along with
a wide NMR band for 31P nuclei with T2 =
25.5 s . Its appearance is indicative of the influence of rotary
averaging of the interaction between magnetic moments of phosphorus
nuclei and protons that is related to the emergence of lateral
mobility of lipid molecules within the membrane [7, 8].
The character of changes in the dynamics of polar regions of Rb. sphaeroides membranes at high values of relative humidity, leaving out of account the absolute values, is on the whole close to the data characterizing the R. rubrum membranes [3, 4]. At the same time, in the region of low and intermediate P/Po values a completely opposite humidity dependence of T1 for phosphorus nuclei was revealed in membranes of R. rubrum bacteria, and there was no left-side symmetry of the resonance form for 31P nuclei (Fig. 2) [3, 4]. These data should also be compared with a relatively low mobility of lipid tails observed in the R. rubrum membranes at low humidity that is specifically suggested by the results of comparison of T2 values for the two types of bacterial membranes under these conditions [1, 2]. In the case of the supposed hexagonal phase, one may expect a relatively loose packing and higher mobility of lipid tails that seem in fact to occur in the membranes of E. shaposhnikovii and Rb. sphaeroides.
Although all these results are consistent with the suggested existence of hexagonal phase for lipids in membranes of Rb. sphaeroides at low humidity, this suggestion stands in need of further evidence. On the other hand, the data obtained for the R. rubrum membranes do not support the occurrence of such phase.
"The differences revealed in the hydration curves and other
structural-dynamic parameters of the three types of photosynthetic
membranes are also associated with noticeable distinctions in
the functional characteristics of these membranes depending on
the relative humidity levels. Incidentally, in the membranes of
R. rubrum a sharp rise of water sorption at P/Po=
0.3-0.5 induces a dramatic increase in the efficiency of electron
transport N in the systems of quinone acceptors (Fig. 3a)
which, similarly to the hydration growth in this range, is more
distinctly pronounced in the RC-LDAO system [1]. This influence
of dehydration on the electron transport efficiency (N) is comparable
with the effect induced in hydrated membranes by the temperature
decrease by about 50°C. However, no such relationship has been
found in the two other types of studied membranes. The decrease
in the electron transport efficiency with the temperature lowering
in all the membranes studied is not necessarily related to the
effect of dehydration. Thus, after dehydration of Rb. sphaeroides
membranes the factor N preserves its high values and exhibits
only a gradual growth with higher humidity approaching 1 at P/Po>
0.6 (Fig. 3b).
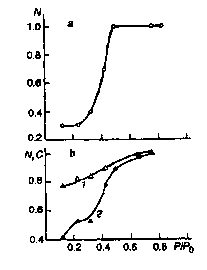
Figure 3. Effect of relative humidity P/P0
of incubation medium on the
efficiency of electron transport between the first and second
quinone acceptors in chromatophores of R. rubrum (a), Rb.
sphaeroides (b, curve 1) and the efficiency of photoinduced
oxidation of high-potencial cytochrome c in chromatophores of
E. shaposhnikovii (b, curve 2).
Similar results were also obtained for the membranes of E. shaposhnikovii [12]. However, in these membranes a different process is observed to occur at P/Po > 0.3 - a dramatic rise of the efficiency of cytochrome photooxidation with its slower subsequent growth at high humidity levels (Fig. 3b). The next stage, consisting in the electron transfer from cytochrome to bacteriochlorophyll in RC, occurs only at high humidity levels (P/Po) = 0.5-0.88), when noticeable changes in this process efficiency take place in the membranes of E. shaposhnikovii bacteria [12]. The dynamics has also a role to play in the recombination of oxidized bacteriochlorophyll with the reduced primary quinone, this process rate being being markedly changed at P/Po > 0.12. However, according to our results, such changes are independent of the lipid phase in the RC of R. rubrum membranes, too - they display the same character when both LDAO and Triton X-100 are used as detergents.
On the whole, the results obtained are indicative of the existence of certain correlation between the structural-dynamic characteristics of photosynthetic membranes and their functional parameters. At the same time, such a correlation is of a complex character revealing a number of differences in membranes of the three bacterial species studied. But the existence of such differences offers additional possibilities for the investigation of these processes making it possible to pass from revealing correlational relationships to considering specific mechanisms and types of interactions which may be responsible for the observed changes and high efficiency of primary photosynthetic processes.
In the first place, let us examine the peculiarities of the biological membrane hydration per se. The above results suggest a number of inferences not only pertinent to both specific membrane types but also bearing a more general character. In the process of membrane hydration, besides common effects related to the polar phase appearance affecting the orientation and localization of lipid molecules, a few stages may be distinguished which seem to differ not merely by the sequence of filling different membrane sites with water but also by the character of structural and dynamic changes induced by such sequential hydration and the effect of these changes on the functional characteristic-s of membranes and their individual components.
The initial steps should involve primarily hydration of polar groups in proteins and lipids. However, in such multicomponent systems as real biological membranes, one should take into account the peculiarities of this hydration. According to the studies of the system protein - low molecular ligands, not all the polar groups of molecules and macromolecules may be hydrated at low humidity because of the interaction of oppositely charged groups of protein macromolecule and ligand [22]. Such an interaction remains virtually unaffected as long as the this system is under the action of bound water with low dielectric constant . As a result, at low and intermediate levels of relative humidity the number of hydrated groups is even smaller than that in an individual protein [22]. The effect of this factor may become particularly essential for biological membranes in which a substantial role is played by the interaction of opposite charges of lipid molecules between themselves in the polar membrane region and with charged groups of proteins. This factor should also influence phase transitions of lipids in the process of membrane hydration.
The next hydration stage which affects markedly the dynamics and functional characteristics of membranes at P/Po = 0.3-0.5, along with a certain increase in the hydration of polar groups, is associated with the insertion of water molecules into hydrogen bonds at some sites of protein globules, as well as between the lipid molecules forming such bonds. This effect is especially pronounced in the R. rubrum membranes with PE predominance in their lipid phase and is weaker in the Rb. sphaeroides membranes with lower PE levels. The effect of substitution of direct hydrogen bonds between lipid molecules and within the protein macromolecules by the bonding via water molecules is observed at smaller P/Po values than those for D2O forming stronger hydrogen bonds compared to H2O. This explains a noticeable prevalence of the heavy water quantity over H2O sorbed at low and intermediate levels of relative humidity in the E. shaposhnikovii membranes, whereas their respective quantities are nearly the same at high humidity (Fig. 1b).
The process of water molecule insertion into hydrogen bonds is also dependent on the water quantity and peculiarities of lipid and protein packing, as indicated by the studies of the effects of water molecule incorporation into the interchain hydrogen bonds on model systems of poly-ethyleneimines [CH2CH2NH]n, [23]. These studies have revealed that this process occurs only in the presence of certain minimal water quantity because the insertion of its molecules into the cross-linking bonds may also result in possible breaks of neighbouring direct bonds. This process becomes advantageous solely with the involvement of a number of water molecules. The effect of packing specifics was also revealed in lipid systems where sharp differences in hydration curves were observed for pure PE and its mixture with phosphatidylcholine. In contrast to its mixture, the pure PE does not virtually sorb water at low and intermediate levels of relative humidity [24].
The effects of water insertion into the cross-linking hydrogen bonds are weak in the case of hydration curves of globular proteins due to a small contribution of such water molecules to the total hydration curve. However, they have significant biological implications which are discussed below.
The third hydration stage, which also has certain specific features and affects noticeably the membrane structure and dynamics, is observed at P/Po> 0.7. Here, besides the general sorbed water increment, there occur additional effects related to the appearance of free water with high dielectric constant . Specifically, this is demonstrated by a considerably larger quantity of sorbed water in the protein-ligands system than in individual proteins, in contrast to the above-mentioned effects at lower humidity levels [22]. As a result, this makes possible hydration of virtually all charged groups which is in turn conducive to the appearance of lateral mobility in the lipid molecules. The latter effect is revealed not only by the drop of T1 values for 13C and 31P nuclei but also by the variation of T2 for a definite portion of 31P nuclei in the studied membranes of R. rubrum [3, 4] and Rb. sphaeroides(Table 2)
Finally, a high, normal for cells, hydration creates conditions allowing regulation of biological processes at the level of cell and individual organelles. In this situation, the participation of water molecules establishes close-to-equilibrium conditions between the bound and free states of proteins, the shift of which may induce, specifically, transitions of weakly bound peripheral proteins to the water medium and back [25, 26]. Noticeable changes in the number of freedom degrees for the protein groups and the lipid molecules, and, respectively, entropy change in such processes should induce a slight free energy change and high sensitivity of such regulation system to pH modification and other impacts. These inferences are supported by a number of direct and indirect data [26]. Furthermore, the concept of reversible transitions of proteins to the water phase allows interpretation of nearly all data known to date on changes in the cytoplasm viscosity, light scattering and intracellular distribution of dyes which accompany the activation of metabolic processes [27, 28].
The above effects of hydration have their distinctive features in various types of membranes depending on their composition and structure, which are revealed by the examination of structural-dynamic and functional peculiarities of the three membrane types studied.
A compact packing of molecules in the polar region and a looser packing of lipid tails in the membranes of Rb. sphaeroides and, apparently, E. shaposhnikovii determine the maintenance of high mobility in the nonpolar regions of dried membranes. This in turn maintains high efficiency of electron transport N in the system of quinone acceptors
QA-QB QAQB- close to 0.7 in the first membrane type and about 1 in the second type. A different situation takes place in the R. rubrum membranes having no such phase. In this system the lipid tails are rather densely packed at low humidity that leads to a dramatic decrease in their mobility and respectively their electron transport efficiency upon drying.
The first hydration stage involving a portion of polar groups seems to induce only local changes in membranes, whereas the next stage determined by the effects of water molecule insertion into hydrogen bonds in membranes has a strong impact on their structure, dynamics and functions. In the R. rubrum membranes, the insertion of water molecules between the PE molecules prevailing in their lipid phase leads not only to a certain ordering of bonds in the polar membrane region but also to a more ordered distribution of lipid molecules and a larger volume of polar phase. This results in a looser packing of lipid tails because of a dramatic rise of their mobility [1,3,4]. This is in turn conducive to a drastic increase in the electron transport efficiency in the system of quinone acceptors. This effect is weaker in the membranes of Rb. sphaeroides and E. shaposhnikovii in which the mobility of lipid tails was already sufficiently high.
The effect of mobility on the efficiency of electron transport in the system of quinone acceptors in the nonpolar phase is determined by its effect on the processes of electrostatic stabilization of reduced quinones at the expense of changes in the distance between charges in the vicinity of QB. This reduces the probability of reverse reaction: to bring it down to 1% and below, it is necessary that the characteristic times of such motions were at least two orders of magnitude smaller than the electron life-time in the resultant state. These requirements are met by the motion times in the nonpolar membrane region which fall into the nanosecond range, as directly indicated by the results of T1 minimum recording for 13C nuclei from the lipid component of R. rubrum membranes [3, 4] as well as the T1 minimum for protons in the E. shaposhnikovii membranes [2] that points to the closeness of motion and resonance frequencies [2]. At the same time, this stage of electron transport is characterized by the times falling in the microsecond range [29].
The insertion of water molecules into hydrogen bonds within the protein limits is less evident from quantitative parameters. Nonetheless, this process seems to actively influence the formation of optimal configuration of proteins, cytochrome c (at least as regards the optimization of functional activity of high-potential haems), as well as the cytochrome-photooxidizing RC protein in membranes of E. shaposhnikovii bacteria. These changes also affect the inner dynamics of proteins, as indicated by a noticeable decrease of T1 values for 13C nuclei in the protein component of R. rubrum membranes [3,4] which is observed with the growing relative humidity. The dynamics in turn makes possible changes in the protein conformation in the course of cytochrome photooxidation which seems to affect the process efficiency. All these changes are observed in the same humidity range where there occurs a dramatic increase of water sorption by the lipids capable of forming hydrogen bonds between their molecules. And here again, as shown by the data reported in [30], the occurrence of enzymic reactions in the solid phase becomes possible, the rates of which reactions prove to be strongly dependent on relative humidity. It is noteworthy that for the RC protein too some data have been reported on the effect of conformational regulation on the the electron transport efficiency, as evidenced by considerable differences in the effective values of rate constants of electron transport under illumination and in the dark derived from the kinetic studies of redox-transformations of RC components based on the identification of parameters of mathematical models [31].
The next hydration step, which also affects substantially the functional characteristics of membranes, is related to the appearance of water with a high dielectric constant . As a result, the interaction between oppositely charged groups of proteins and lipids is weakened thus making possible lateral difftision of lipid molecules and, apparently, proteins within the membranes. Owing to changes in the distance between the interacting structures, the lateral diffusion seems to influence the above-mentioned variation of the efficiency of electron transport from cytochrome c to bacteriochlorophyll of RC which is observed at this hydration stage of photosynthetic membranes of E. shaposhnikovii [2]. Specifically, such a process should prevent possible structural perturbations by the unused electron flux, whereas diffusion will also affect the regulation of photo-synthetic processes at the level of mobile carriers. An important role of medium properties, such as its viscosity and pH, in the regulation of photosynthetic reactions was demonstrated on mathematical models of primary processes of photosynthesis [31].
In other membrane types of photosynthetic purple bacteria cytochromes c are bound more weakly than in membranes of E. shaposhnikovii and are known to be readily separated from membranes in the process of isolation of chromatophores under the increasing ionic strength. The extent of binding of these cytochromes to membranes appears to vary during functioning of photosynthetic membranes that, as noted above, must be related to a relatively small variation of free energy but to a sufficiently long impact (required for overcoming the activation barrier) [26]. This may serve the purpose of another factor in regulating the efficiency of photosynthetic processes and preventing possible structural perturbations of these membranes, as well as organelles and the entire cell.
Thus, comparative investigation of the effects of hydration in three types of photosynthetic membranes of purple bacteria made it possible to reveal a number of peculiarities in the structure of such membranes and mechanisms of its modification in the process of hydration. It has been shown that the hydration of biological membranes does not boil down to a certain sequence of filling specific sites of the surface of macromolecules or lipid phase but is associated with profound changes in their structural and dynamic characteristics. The entire process may be divided into four stages of hydration effect. The first stage includes local changes in the structure and dynamics of membranes with possible shifts of individual groups to distances on the order of atomic sizes. The next hydration stage with insertion of water molecules into hydrogen bonds in membranes leads to a large-scale structural rearrangement, involving protein macromolecules and a considerable portion of the lipid phase. The scope of motion of each individual group with frequencies of the order of 109 s-1 appears to preserve its local character. Though as a result of motion of numerous groups, the conformation of proteins and the state of some lipid phase sites undergo noticeable changes during longer time intervals. And only at the third stage the appearance of water with high dielectric constant makes possible mutual displacements of membrane components to distances larger than atomic sizes. This stage is characterized by a noticeable increase of the sensitivity of electron transport along the photosynthetic chain to various regulatory impacts. Finally, at the fourth stage with conditions for normal hydration extent, the electron transport processes become still more sensitive to regulation, on the whole. All this points to a close interrelation of hydration processes and the character of inner motion in membrane structures which is a crucial factor for achieving high efficiency of electron transport at different sites of the photosynthetic chain and optimization of conditions of its functioning.
This work was in part supported by the International Science Foundation and the Government of the Russian Federation (grant J 16100) and the Russian Foundation for Basic Research (grant 95-04-1857a).
REFERENCES